Summary: A validated model to test the anti-angiogenic potential of a drug.
Model Description
This model is used to test the anti-angiogenic potential of an active pharmaceutical ingredient (API) in human umbilical vein endothelial cells (HUVEC) in vitro.
HUVECs are grown on 15-well µ-Slide Angiogenesis (Ibidi GmbH, Germany). The cells can be exposed to study compounds at different concentrations for 6h.
| Cells | Human umbilical vein endothelial cells (HUVEC) |
| Induction | Angiogenin and VEGF |
| Positive control | Sulforaphane |
| Analysis | ImageJ or Wimasis |
| Read-outs | Total tube length, total number of segments |
Outcomes and Read-Outs
Imaging
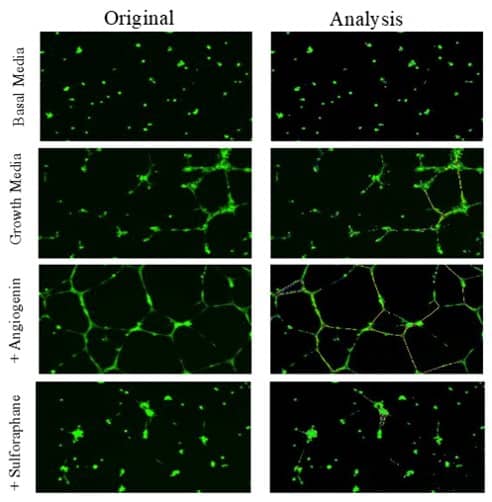
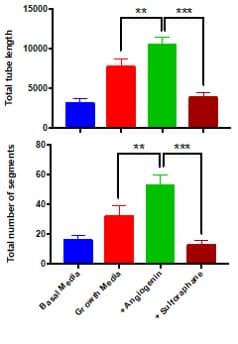
References
- Park HJ, Zhang Y, Georgescu SP, Johnson KL, Kong D, Galper JB. Human umbilical vein endothelial cells and human dermal microvascular endothelial cells offer new insights into the relationship between lipid metabolism and angiogenesis. Stem Cell Rev. 2006;2(2):93-102. Review. PubMed PMID: 17237547.
- Cao Y, Gong Y, Liu L, Zhou Y, Fang X, Zhang C, Li Y, Li J. The use of human umbilical vein endothelial cells (HUVECs) as an in vitro model to assess the toxicity of nanoparticles to endothelium: a review. J Appl Toxicol. 2017 Dec;37(12):1359-1369. doi: 10.1002/jat.3470. Epub 2017 Apr 6. Review. PubMed PMID: 28383141.
- Khoo CP, Micklem K, Watt SM. A comparison of methods for quantifying angiogenesis in the Matrigel assay in vitro. Tissue Eng Part C Methods. 2011 Sep;17(9):895-906. doi: 10.1089/ten.TEC.2011.0150. Epub 2011 Jun 8. PubMed PMID: 21517696.



